Efficacy evaluations included inflammatory and noninflammatory lesion counts and EGSS at screening, baseline, and during treatment (weeks 4, 8, and 12).10 Primary efficacy end points included absolute change in mean inflammatory and noninflammatory lesion counts and the proportion of patients who achieved at least a 2-grade reduction in EGSS from baseline to week 12 (treatment success at end of study). Secondary efficacy end points included mean percentage change from baseline to week 12 in inflammatory and noninflammatory lesion counts and the proportion of patients who considered themselves clear or almost clear at week 12.10
After 12 weeks of daily treatment, inflammatory and noninflammatory lesion counts decreased by a mean of 60.4% and 51.8%, respectively, with clindamycin-BP 3.75% gel compared to 31.3% and 27.6%, respectively, with vehicle (both P<.001). At weeks 4, 8, and 12, the difference in inflammatory and noninflammatory lesion counts for the active treatment was 17.4%, 24.8%, and 29.1%, respectively, and 8.1%, 19.8%, and 24.2%, respectively, for vehicle.10
Treatment success (at least a 2-grade improvement in EGSS) was achieved by 9.1% of patients using clindamycin-BP 3.75% gel compared to 4.6% using vehicle by week 4. Additionally, 6.3% of patients considered their AV as clear or almost clear compared to 3.5% with vehicle at week 2 (Figure 1).10
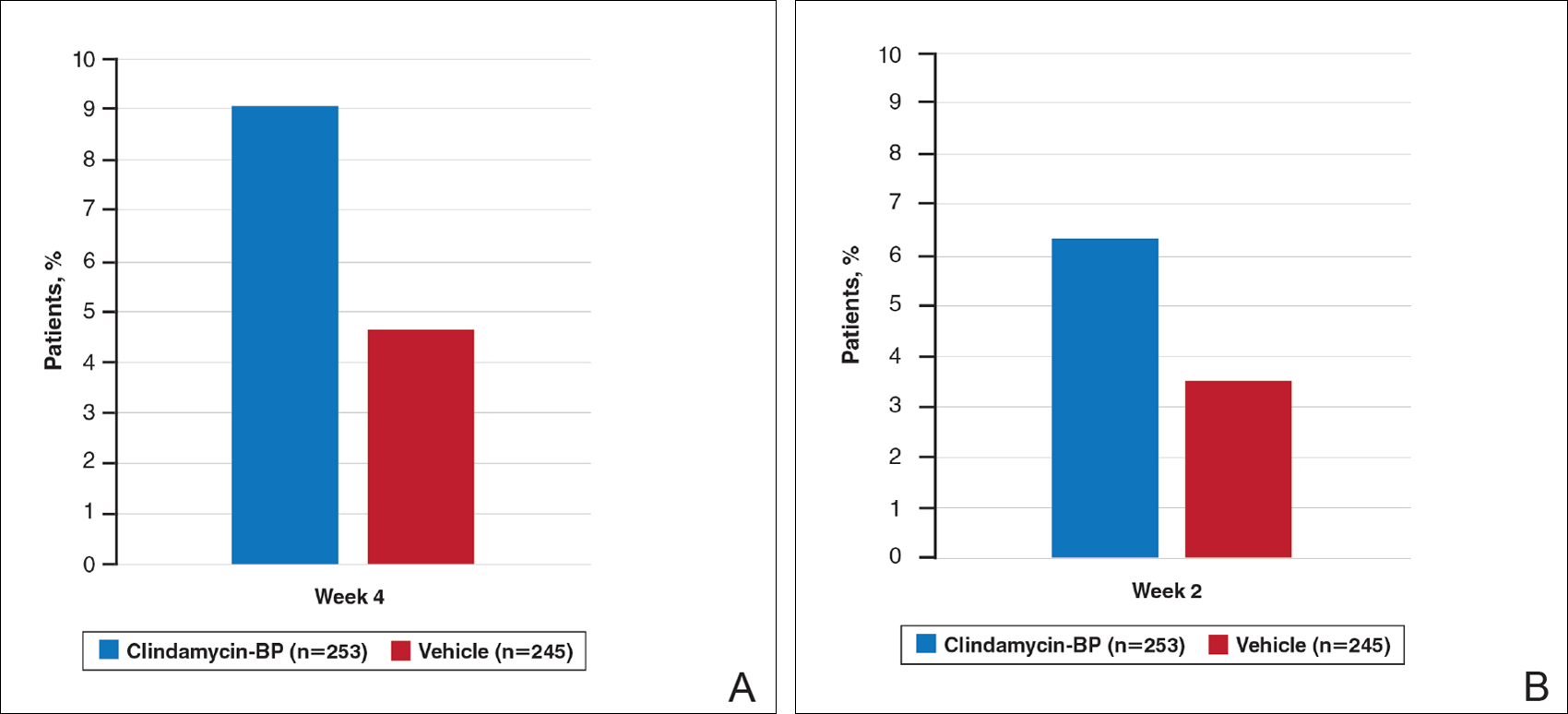
Figure 1. Number of patients considered treatment successes within 2 to 4 weeks using investigator (A) and patient self-assessment (B). Investigators reported treatment success as patients achieving at least a 2-grade improvement in Evaluator’s Global Severity Score. Patients reported treatment success as acne that was clear or almost clear. BP indicates benzoyl peroxide. Data from Pariser et al.10
This analysis represents the first attempt to evaluate and report TOA results with clindamycin-BP 3.75% gel. Time to onset of action for inflammatory lesions treated with clindamycin-BP 3.75% gel was calculated as 2.5 weeks versus 6.2 weeks for vehicle (Figure 2A). Time to onset of action for noninflammatory lesions was 3.7 weeks with clindamycin-BP 3.75% gel versus 8.6 weeks with vehicle (Figure 2B). The difference in TOA between the active and vehicle study groups was 3.7 weeks and 4.9 weeks, respectively. In addition, among actively treated patients, TOA was shorter in females (2.1 weeks) than in males (2.6 weeks) and in moderate AV (2.5 weeks) compared to severe AV (3.0 weeks).
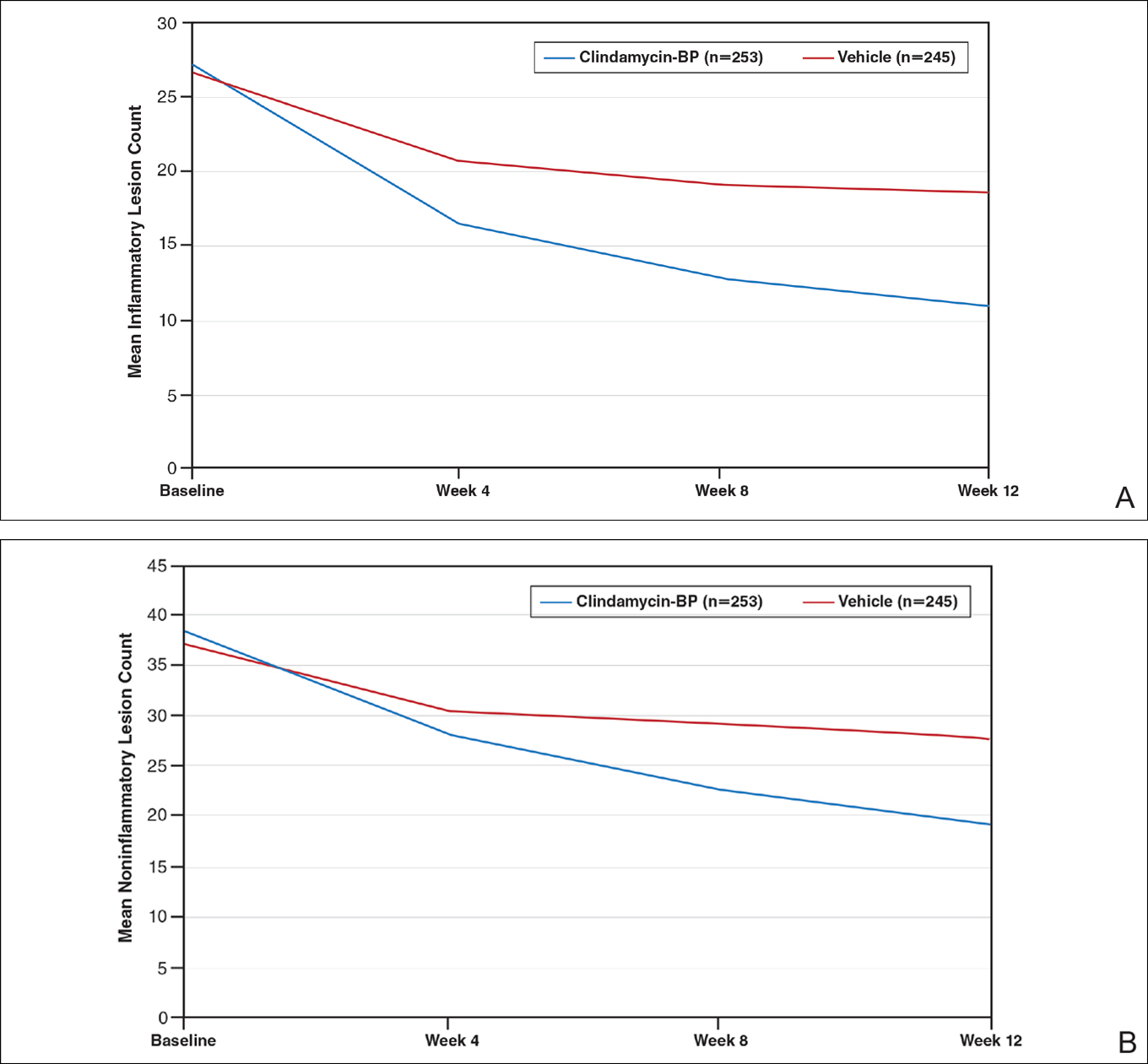
Figure 2. Inflammatory (A) and noninflammatory (B) lesion count reduction with clindamycin–benzoyl peroxide (BP) 3.75% gel and vehicle from baseline to week 12 (intention-to-treat population). The clindamycin-BP 3.75% gel mean inflammatory lesion count at baseline was 27.4; 15% reduction at 1.4 weeks; 25% reduction at 2.5 weeks; and 50% reduction at 7.2 weeks. The vehicle mean inflammatory lesion count at baseline was 26.7; 25% reduction at 6.2 weeks; 50% reduction not achieved. The clindamycin-BP 3.75% gel mean noninflammatory lesion count at baseline was 38.3; 25% reduction at 3.7 weeks; and 50% reduction at 11.9 weeks. The vehicle mean noninflammatory lesion count at baseline was 37.2; 25% reduction at 8.6 weeks; 50% reduction not achieved.
Comment
Differences in lesion counts between clindamycin-BP 3.75% gel and vehicle suggest a clinically relevant benefit in favor of active treatment with both inflammatory and noninflammatory lesions. Nearly twice as many patients were rated as treatment successes using EGSS by week 4 or clear or almost clear as early as week 2 compared to the vehicle group.10 However, these data are suggested as an overall guide but do not provide adequate guidance on when visible improvement may start to be evident in a given patient.
The analysis reported here shows a TOA of 2.5 weeks with clindamycin-BP 3.75% gel for inflammatory lesions, approximately 4 weeks faster than with the vehicle. In most cases, a reduction in inflammatory lesions is more likely to have a greater impact on patient perception of TOA. Unless a patient is aware or focused enough to actively distinguish visibly between inflammatory and noninflammatory (comedonal) AV lesions, their eye is more likely to be drawn initially to reduction in inflammatory lesions, which are erythematous and more visible at a greater viewing distance. Although noninflammatory AV lesions usually require closer inspection to visualize them (especially closed comedones), they are often slower to respond to treatment. Analysis of the pivotal trial data reports a longer TOA with clindamycin-BP 3.75% gel for noninflammatory lesions (3.7 weeks) versus inflammatory lesions (2.5 weeks).
As expected, TOA was shorter in patients with moderate AV than severe AV (2.5 weeks vs 3.0 weeks). Time to onset of action also was shorter in females overall. It is unclear why we see gender differences in acne studies. A number of reasons have been suggested, including differences in AV pathophysiology and/or treatment adherence.11,12 Greater efficacy of clindamycin-BP 3.75% gel in females compared with males has already been reported, and better overall efficacy leading to a shorter TOA has been noted by others.13

